Abstract
Background: Myelofibrosis (MF) is characterized by driver mutation (JAK2, CALR and MPL) mediated hyperactivity of the JAK-STAT signaling pathway, bone marrow fibrosis, impaired hematopoiesis, and splenomegaly /extramedullary hematopoiesis. Ruxolitinib (rux), a JAK inhibitor (JAKi), is the standard of care for higher risk MF patients. In 2019, the US FDA approved Fedratinib, a second JAKi that binds to both the ATP and substrate binding sites of the JAK2 kinase domain. The phase II JAKARTA2 study demonstrated that fedratinib is clinically efficacious in previously rux exposed patients. In the current work, through biomarker analysis of the phase 3b FREEDOM data, we sought to identify changes to the cytokine profile in fedratinib treated, previously rux exposed patients. In addition, we explored potential mechanism of fedratinib efficacy.
Methods: In the FREEDOM study, patients (N=38) received fedratinib (400 mg/day) once daily in 28-day cycles until lack of efficacy, intolerance, or disease progression. Primary endpoint was proportion of subjects with ≥35% reduction of spleen volume assessed by magnetic resonance imaging (MRI) at the end of cycle 6 (SVR35). In the FREEDOM biomarker cohort, mutational detection was performed in immunomagnetically-enriched CD66+ myeloid cells using the MLL myeloid panel targeting 74 genes. Variants and indels were called using Pisces and Pindel, followed by annotation to COSMIC, ClinVar, gnomAd (non-cancer), and dbNSFP data bases. Clinically relevant somatic mutations were considered for analysis. Rules-Based Medicine's (RBM) HumanMAP v2.0 panel targeting 85 cytokines was used to measure changes in serum cytokine levels. 51 cytokines with variance > 0.3 on aggregated data were used for analysis. Pearson correlation analysis was used to compute Rho values between response and cytokine changes. Kinome screen was performed using the thermofisher Lanthascreen Eu kinase binding assay and the reaction biology kinome assay.
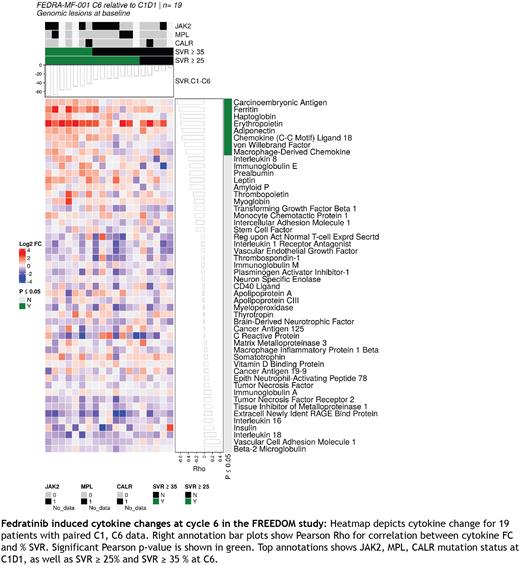
Results and discussion Therapy related disease modifying effect in MF is currently being explored through ability to control variant allele frequency (VAF), reduce inflammation, minimize bone marrow fibrosis, in conjunction with restoration of normal hematopoiesis. In our study, JAK2 V617F mutation was detected in 48% (N=13/27) of total mutational biomarker cohort (N=27/38). However, response was independent of JAK2 V617F mutational status thereby decoupling this mutation with response/disease modification at cycle 6. As a next step, we evaluated if fedratinib treatment results in cytokine alterations that correlate with clinical response. Cytokine evaluation and paired analysis between cycle 1 and end of cycle 6 revealed increases in adiponectin, carcinoembryonic antigen (CEA), EPO and ferritin and decrease in EN-RAGE, IL-16, IL-18, IL1RA, TIMP-1, TNFR2, MPO, VCAM1 and VEGF (P < 0.001). Correlation analysis revealed adiponectin, CEA, ferritin, haptoglobin, erythropoietin and CCL18 to have significant correlation (P < 0.05) with response. The responders had the largest alteration in cytokine levels coupling these cytokines to response. Overall, fedratinib upregulates anti-inflammatory cytokines and reduces inflammatory cytokines which could contribute to symptom burden control and spleen volume reduction. These results in a rux exposed patient cohort suggests that fedratinib could have a differentiated profile from rux. To further investigate these differences, we performed kinome screens with two in vitro assays. Results indicate that fedratinib has a broader kinase inhibitory profile than rux. Thus, fedratinib could modulate additional signaling pathways explaining potential efficacy in rux exposed patients.
Conclusion: Our biomarker analysis in previously rux exposed patients, shows that fedratinib triggers cytokine changes that correlate with response independent of mutational status. This suggests that at least at early timepoints, cytokine changes could be a potential biomarker for disease modifying effect. In addition, our in vitro analysis differentiates rux and fedratinib based on the inhibited kinases and altered biological pathways. Taken together our results indicate that while both rux and fedratinib are JAK inhibitors, they are well differentiated allowing fedratinib to be effective in previously rux exposed patients.
Disclosures
Jeyaraju:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company, Patents & Royalties: Methods of treating cytokine-related adverse events. Hayati:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Polonskaia:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company, Research Funding. Alapa:Bristol Myers Squibb: Current Employment. Ugidos:BRISTOL MYERS SQUIBB: Current Employment. Browne:Bristol Myers Squibb: Current Employment, Research Funding. Risueño:Bristol Myers Squibb: Current Employment, Current equity holder in private company, Divested equity in a private or publicly-traded company in the past 24 months, Patents & Royalties. Hagner:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Gupta:Roche: Other: Participation on a Data Safety or Advisory board; Novartis: Consultancy, Honoraria; Constellation Pharmaceuticals, Inc., a MorphoSys Company: Consultancy, Honoraria; AbbVie: Consultancy, Other: Participation on a Data Safety or Advisory board; BMS Celgene: Consultancy, Honoraria, Other: Participation on a Data Safety or Advisory board; Sierra Oncology: Consultancy; Pfizer: Consultancy, Other: Participation on a Data Safety or Advisory board. Talpaz:IMAGO: Consultancy; Kirin: Membership on an entity's Board of Directors or advisory committees; SDp: Membership on an entity's Board of Directors or advisory committees; Takeda: Other: Grant/research support ; Novartis: Consultancy, Other: Grant/research support ; BMS: Consultancy. Hernandez:Bristol Myers Squibb: Current Employment. Chia:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company, Other: Travel. Martin-Regueira:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Lopes de Menezes:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company, Patents & Royalties. La Motte-Mohs:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company; MacroGenics: Current equity holder in publicly-traded company, Patents & Royalties: Patents; Notch Therapeutics: Patents & Royalties: Patents. Suragani:Acceleron pharma: Ended employment in the past 24 months; Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Gandhi:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal